Firefly / Lightning Bug
Photuris and Photinus species
New research uses the firefly’s light-emitting chemicals in determining how general anesthetics work. Live adult fireflies photographed at Hidden Lake Forest Preserve, Glen Ellyn, Illinois.
 More images at PLoS [4] |
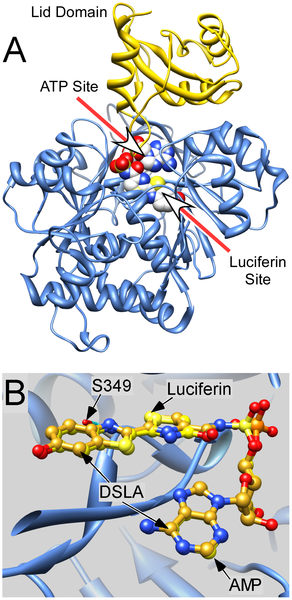
Despite many years of effort, there is no scientific consensus as to how general anesthetics act, or even the molecular nature of their target sites in the human central nervous system. Most attention has been focused on two extreme views: anesthetics act by binding directly to protein molecules, or they exert their effects by disrupting the lipid portions of nerve mem-branes [3]. In an effort to pinpoint the exact nature of the binding site interactions, scientists are using the light-emitting properties of chemicals found in fireflies to determine the affinity of variously *photolabeled chemicals in inhibiting light emission, and hence the nature and location of the binding sites on the protein. Due to its high sensitivity and extreme specificity for ATP, luciferase is used as a marker in the investigation of various biological processes; an investigation into the color of the firefly’s light suggests that the wavelength depends on the polarity as influenced by the microenvironment inside the luciferase protein cavity [5] . Firefly luciferase is one of the few soluble proteins that is acted upon by a wide variety of general anesthetics and alcohols; they inhibit the ATP–driven production of light. Scientists have used time–resolved photolabeling to locate the binding sites of alcohols during the initial light output, some 200 ms after adding ATP. The photolabel 3-azioctanol inhibited the initial light output with an IC50 of 200 µM, close to its general anesthetic potency. Researchers used recombinant luciferase derived from two different species of firefly, Luciola cruciata (Japan), and North America’s Photinus pyralis, to determine how general anesthetics interact with protein molecules by locating the anesthetic sites of action on firefly luciferase during the initial few hundred milliseconds of ATP–induced light emission [4]. Photolabeled luciferase, along with luciferin and differing concentrations of the alkanol 3-azioctanol were rapidly mixed with ATP in a stopped-flow spectroflourimeter, and the resulting burst of light for each condition was recorded; the slope of the linear region of the curve was used to characterize the level of the luciferase activity. Increasing concentrations of 3-azioctanol inhibited the initial phase of light emission [4]. The nature and location of photolabeled residues gives the scientists a better understanding of how anesthetics interfere with the binding of ATP and luciferin into the luciferase molecule (along with the resultant inhibition of photon emission); the results may help locate similar binding sites in our own nervous system proteins and accelerate the development of cheaper, safer, and more environmentally-friendly anesthetics than currently in use. |
*Photolabeling, shorthand for Nucleotide photoaffinity labeling, is a powerful tool of research in the biological sciences.
“Nucleotides and nucleotide binding proteins regulate numerous biochemical processes in the cell. Many of the nucleotide binding proteins controlling these biochemical process are are the targets of drugs as well as toxicants. In addition, many of these nucleotide binding proteins are affected or altered in various disease states. The use of nucleotide photoaffinity analogs allows for the study of protein-nucleotide interactions at the molecular level.
This powerful technique allows one to identify the amino acid residues actually involved in formation of the active site of nucleotide binding proteins and how mutations in that active site subsequently affect nucleotide binding. It also allows one to study how substrates, cofactors, drugs, toxicants, etc. all affect binding of nucleotides to the active site of proteins. The information gained from these studies allows researchers to determine how these nucleotide binding proteins regulate the biochemical processes in which they are involved. In addition, this technique can be used to determine which specific nucleotide binding proteins are the targets of drugs and toxicants as well as how these proteins are regulated.” — Affinity Photoprobes LLC
Fireflies (we called them “lightning bugs” as kids) are neither flies nor bugs; they are beetles. Because of the crepuscular nature of the insect itself, its lackadaisical flight pattern, and the luminous organ at the tip of the abdomen (they are, after all, advertising their presence), fireflies are some of the most familiar, and surprisingly well-liked (perhaps I should less-feared) insects. It is an incomplete childhood, indeed, that does not contain a summer evening or two spent chasing and capturing these charming little fellows. I shudder to think of the large number of unfortunates that suffered captivity in my youthful Mason jar, brought into a dark bedroom at night to see if I could surreptitiously read comic books by their light (no), or summarily dismembered – more like disabdomened – to be smeared onto our skin as “jewelry.” Primitive human blacklight posters, if you will.
Fireflies use different intermittent signals in order to attract the attention of a mate. Both genders use a specific flash sequence and duration, and each species has a distinct dialect. In the firefly mating world, competition is fierce. It is not unusual for a female of one species to mimic the signals of another to lure a male for the sole purpose of eating him. Males of the species Photuris lucicrescens also mimic other species’ patterns to lure them as prey. So there. They dish it out too, and I don’t feel bad anymore about squashing a few so long ago.
References
- Bugguide.net “Genus Photuris“
- Bugguide.net “Genus Photinus“
- Probing the molecular dimensions of general anaesthetic target sites in tadpoles (Xenopus laevis) and model systems using cycloalcohols. S. Curry, G. W. Moss, R. Dickinson, W. R. Lieb, N. P. Franks Br J Pharmacol. 1991 January; 102(1): 167–173. PMCID: PMC1917901
- Shanmugasundararaj S , Lehle S , Yamodo HI , Husain SS , Tseng C , et al. 2012 | The Location and Nature of General Anesthetic Binding Sites on the Active Conformation of Firefly Luciferase; A Time Resolved Photolabeling Study.
PLoS ONE 7(1): e29854. doi:10.1371/journal.pone.0029854 - Lisa Zyga, Physorg.com, “Scientists Investigate How Fireflies Emit Different Colors”
Beetles Main | Beetles Index | Longhorns | Leaf Beetles | Soldier | Blister | Lady | Scarab

 Firefly Photinus pyralis
Firefly Photinus pyralis Firefly Photuris species
Firefly Photuris species